Adults verses the Kids over the Ukraine War, Adults discuss the menu, Kids are food on the table to be exploited. Kids asked for it, blame no one except own stupidity 成人與孩子在烏克蘭戰爭中的分別, 成人討論菜單,孩子則是餐桌上可供利用的食物。這是孩子們自找的,怪不了別人,只能怪自己愚蠢.


Adults verses the Kids over the Ukraine War, Adults discuss the menu, Kids are food on the table to be exploited. Kids asked for it, blame no one except own stupidity 成人與孩子在烏克蘭戰爭中的分別, 成人討論菜單,孩子則是餐桌上可供利用的食物。這是孩子們自找的,怪不了別人,只能怪自己愚蠢.


Video: 700 Free Na Ji 2 movie tickets sponsored by Alex Yeung for his patrons & real estate clients in HK 香港華記2月18報導:一齊贏華記請您免費看電影「哪吒2之魔童鬧海」呼籲全香港大中小規模公司發揮企業精神免費請員工一齊睇哪吒電影!一齊贏一齊開心,帶動香港內需循環,振興香港經濟。大家一齊行街睇戲食飯搭車購物
https://rumble.com/v6mc6nu-700-free-na-ji-2-movie-tickets-sponsored-by-alex-yeung.html
https://www.tiktok.com/t/ZP8YXwVB1/
https://youtu.be/4f6aDIjm2RE?si=zADg9oI0Jp-gL2Ih


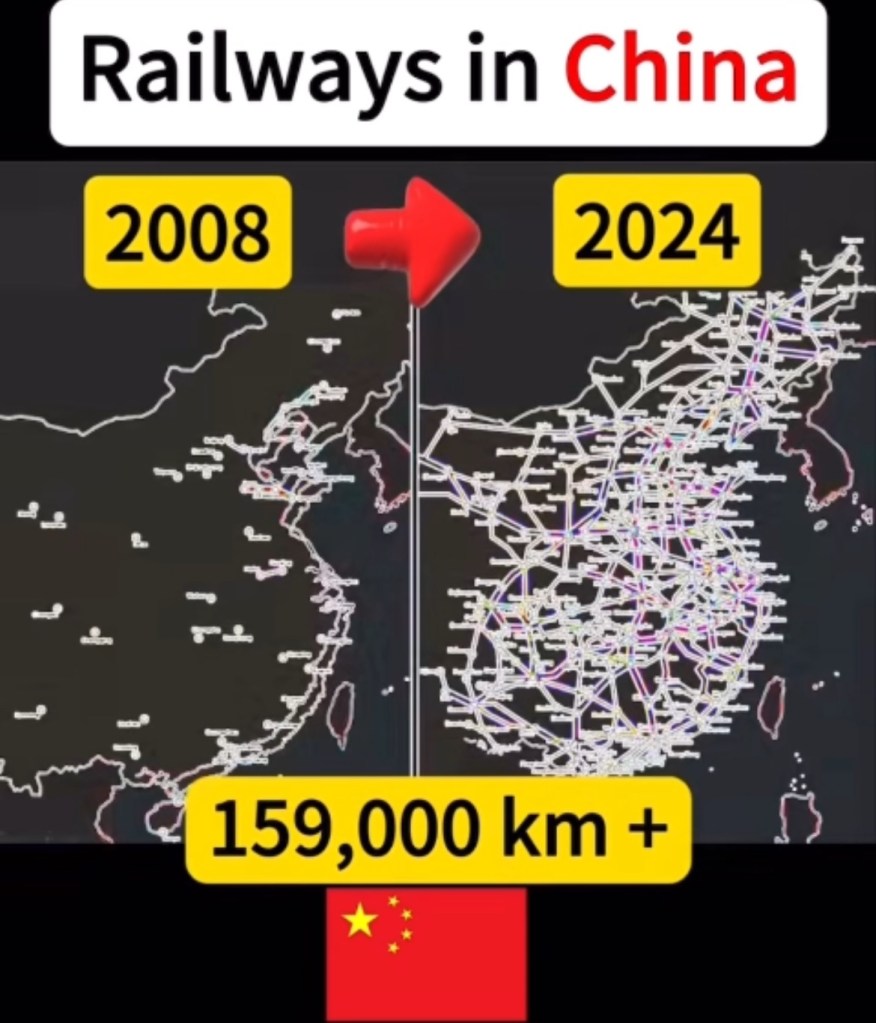
Video: How China Built the World’s Longest Tracks in Record Time with best engineers and engineering techniques! No one could match or beat China! 中國如何以最優秀的工程師和工程技術在創紀錄的時間內建成世界上最長的軌道, 沒有人能夠匹敵或擊敗中國!
https://rumble.com/v6mbd4i-how-china-built-the-worlds-longest-tracks-in-record-time.html
https://www.tiktok.com/t/ZP8Y4dD4g/
https://youtu.be/xZzqjAklWKo?si=RThYrg-BpENDZ5R2
The best of the best Chinese used to come to US! Time has changed especially the last 10 years! The best of the best Chinese leaves US when the future is China, definitely not US. SCMP: Another Smart Chinese Scientist left US for China, Veteran Google AI researcher joins Chinese TikTok owner ByteDance. Wu Yonghui, who worked at Google for 17 years, will lead foundational research at the Seed division and report directly to CEO Liang Rubo, sources say. 從前最優秀的中國人要來美國!時代變了,特別是過去的十年!最優秀的中國人離開了美國,因為世界的未來是屬於中國的,而不是美國! 南華早報:另一位聰明的中國科學家離開美國前往中國,資深谷歌人工智慧研究員加入中國 TikTok 所有者位元組跳動。據悉,曾在Google工作17年的吳永輝將領導種子部門的基礎研究,並直接向執行長樑如波報告.

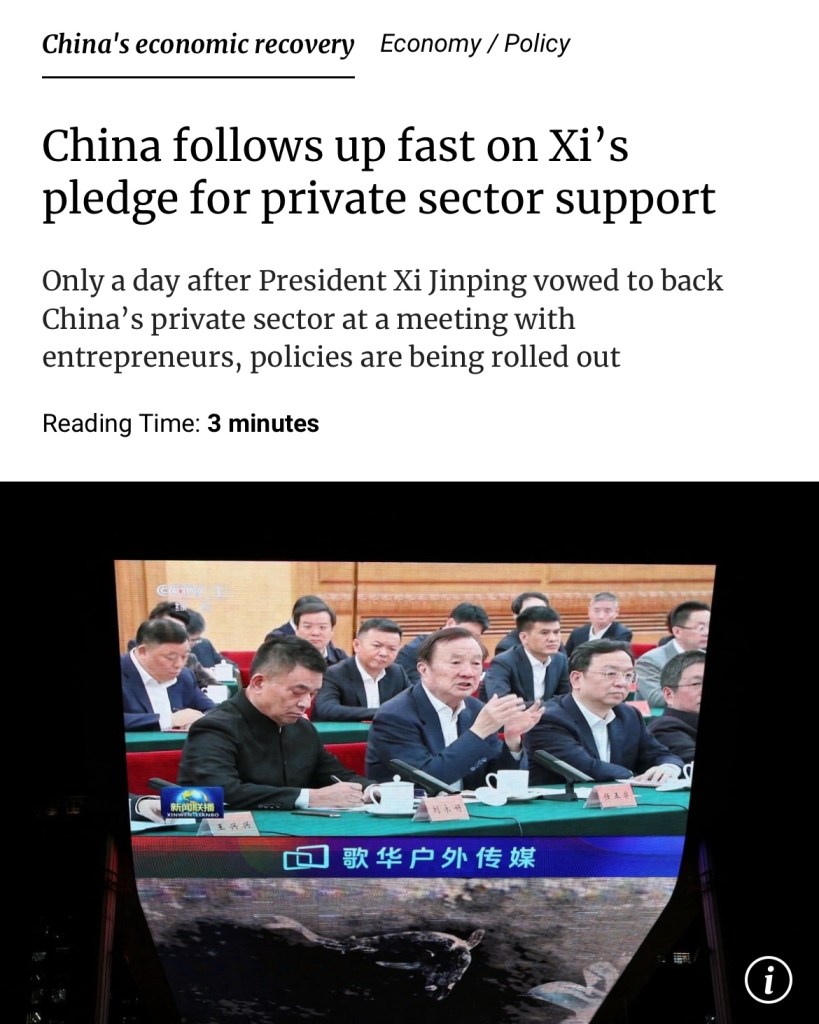
SCMP: One day after a high-profile symposium hosted by President Xi Jinping and attended by China’s leading entrepreneurs, Beijing has unveiled new measures in support of the private sector – an act the president explicitly backed in his speech. 《南華早報》:在國家主席習近平主持召開中國多位著名企業家參加的高調研討會的第二天,北京出台了支持私營部門發展的新措施, 主席在講話中明確支持這一舉措.





The best of the best Chinese used to come to US! Time has changed especially the last 10 years! The best of the best Chinese leaves US when the future is China, definitely not US. SCMP: Another smart distinguished Chinese scientists left US where China is the future for science and technology. After more than two decades of distinguished work in the United States, geophysicist Niu Fenglin has returned to China to join Earth and space sciences school at the University of Science and Technology of China (USTC). 從前最優秀的中國人要來美國!時代變了,特別是過去的十年!最優秀的中國人離開了美國,因為世界的未來是屬於中國的,而不是美國. 南華早報:又有一位聰明的中國科學家離開了美國,中國是科學技術的未來。在美國工作二十多年後,地球物理學家返回中國,加入中國科學技術大學地球與太空科學學院.

Video: Former HK ICAC deputy director analyzes US deep structural corruption & Musk work exposed former US Presidents like Clinton, Obama, Biden and US House Speaker Palosi could cost his life X檔案「香港ICAC前副署長」徐家傑馬斯克比香港廉政公署責任更廣, 要處理美國深層次貪污腐敗即將被暗殺?特朗普平反2019香港黑暴?徐Sir美國50個州即將分裂3分天下.
Complete video: https://youtu.be/t-vrhvpM89c

Taiwan US-China expert video: Trump/Musk to terminate USAID & NED, the leftist organ control by Democrats created chaos in the world like the black terror in HK in 2019 don’t benefit US, Clinton, Obama & especially Nancy Palosi made US$200k salary, how could they worth US$200 millions today? 台灣中美尊家視頻: 川普/馬斯克要終止美國國際開發署和國家民主基金會,民主黨控制的左翼機構給世界造成了混亂,什麼顏色革命,暗殺,到處用民主之名像2019香港黑暴祇是其中之一,民主黨做盡這些壞事卻對美國沒有一點好處,克林頓、歐巴馬,尤其是南希·帕洛西的薪水是 20 萬美元,但他們今天怎麼能有2 億美元財富,錢從哪裡來,一定要查!
https://rumble.com/v6lxp97-trumpmusk-to-terminate-usaid-and-ned.html
https://www.tiktok.com/t/ZP8YCPnnY/
https://youtu.be/mYvy0S7lQ5U?si=spSDO8SLw8wN9n1R
Complete story video: https://www.youtube.com/live/5xpvvk3zBs8?si=QCX5Z9YOtO-k83cj

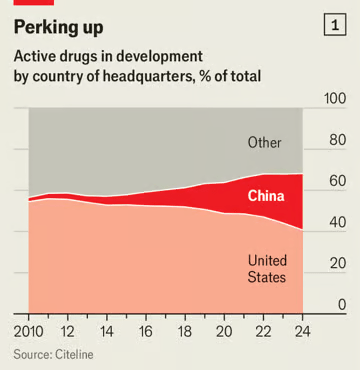
The Economist: It’s not just AI. China’s medicines are surprising the world, too! Its firms are at the forefront of cheaper, faster drug discovery 經濟學人:不只是人工智慧。中國醫藥也讓世界驚嘆!其公司處於更便宜、更快速的藥物研發領域的前沿
eytruda, a cancer-immunotherapy medicine, ranks among the most lucrative drugs ever sold. Since its launch in 2014 it has raked in over $130bn in sales for Merck, its American maker, including $29.5bn last year. In September last year an experimental drug did what none had done before. In late-stage trials for non-small-cell lung cancer, it nearly doubled the time patients lived without the disease worsening—to 11.1 months, compared with 5.8 months for Keytruda.
The results were stunning. So too was the nationality of the company behind them. Akeso, a biotech firm, is Chinese.
In recent months China’s progress in artificial intelligence has stunned the world. A quieter, yet equally significant shift is under way in biotech. China has long been known for churning out generic drugs, supplying raw ingredients and running clinical trials for the pharmaceutical world. But its drugmakers are now also at the cutting edge, producing innovative medicines that are cheaper than the ones they compete with. China has become the second-largest developer of new drugs, behind only America
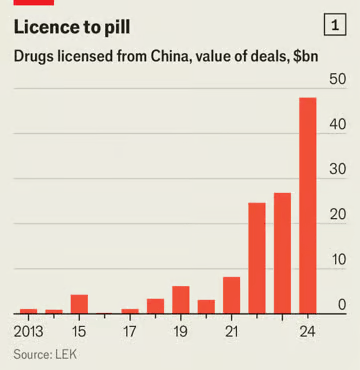
As a consequence, Western drugmakers are increasingly looking east for ideas. Because of expiring drug patents, they stand to lose as much as $140bn a year in sales by the end of the decade. Last year nearly a third of their large licensing deals they struck—those worth $50m or more—were with Chinese firms, triple the share of 2020. lek, a consultancy, estimates that during that time, the total value of drugs licensed in the West from China rose 15-fold, to $48bn (see chart 2). In November Merck paid $588m to LaNova Medicines, another Chinese biotech firm, to secure rights to a therapy similar to that produced by Akeso.
China’s government identified biotech as a strategic priority nearly two decades ago. But it was not until 2015 that things really took off, after the national drug regulator launched ambitious reforms. It took on more staff and cleared a backlog of 20,000 drug applications in just two years. Clinical trials were streamlined and brought into step with global standards. A study by Yimin Cui of Peking University and colleagues, published in Nature Reviews Drug Discovery, found that the time taken to approve the first round of human trials fell to 87 days, from 501 days before the reforms.
The reforms coincided with a wave of returning “sea turtles”, the term for Chinese scientists who had studied or worked abroad. China’s vast domestic market added momentum, by drawing every major drugmaker to its shores, bringing know-how and talent. Easier listing rules gave biotech investors a clearer path to exit, making the sector more attractive. Private funding for Chinese biotechs rose from $1bn in 2016 to $13.4bn in 2021.
With more brains and money, Chinese firms moved beyond copying Western drugs. Instead of waiting for patents to expire and making identical generics, they adopted a “fast-follower” strategy—taking known drugs and modifying them to improve safety, efficacy or delivery. Drug development typically starts by identifying a target, usually a protein or gene linked to a disease. Scientists then search for molecules that can either block or boost the target’s function. Since fast followers are not starting from scratch, they can run trials at a fraction of the cost and speed.
Between 2021 and 2024 the number of Chinese drugs in development doubled to 4,391. Fast-follower and completely original treatments made up nearly 42% of the pipeline. Helen Chen from lek notes that China’s approach has been particularly effective in a type of cancer medicine called adcs, in which an antibody is attached to a payload of chemotherapy via a chemical linker. Since the core components of the treatment already exist, success depends on combining them in the most effective way. Ms Chen believes this is where Chinese firms thrive.
Speed is another advantage, says Michelle Xia, founder of Akeso. “We can do things twice or even three times faster than anywhere else in the world,” she claims. Clinical trials—the longest and most expensive stage of drug development—are faster than in the West (hence why China runs trials for other parts of the world). A large patient population makes recruitment easier, and hospitals and doctors are incentivised by the government to support research.
Faster trials have made Chinese drugs even more attractive to global drugmakers. Though the clinical information is from mostly Chinese patients, rather than a wider sample, it helps investors and pharma firms spot promising treatments. And as the quality of data from China has improved, other regulators are paying attention. Results from Akeso’s Chinese trials were strong enough to convince America’s Food and Drug Administration to move the drug straight into late-stage trials.
Few Chinese firms sell their drugs in America directly. Instead they tend to strike licensing deals: a company sells the rights to market its drug outside China in exchange for an upfront payment, milestone-based fees and royalties on future sales. Akeso’s Keytruda competitor was licensed to Summit Therapeutics, an American biotech, for $500m upfront, with up to $5bn in additional payments and a share of royalties.
Another approach is the “NewCo” model. This involves a Chinese pharma company spinning off its clinical assets into a separate American entity, run by an experienced local management team. The parent firm retains partial ownership, allowing it to benefit beyond royalties if the drug succeeds. Jefferies, an investment bank, estimates that around eight such companies have been formed since May.
China’s biotech boom is not without risks. The rise in licensing deals hides a funding crunch. Private investment in Chinese biotech fell to a seven-year low in 2024, mirroring a slowdown in global biotech markets. Investors are prioritising companies that generate cash or have strong international potential. Jimmy Zhang, an investor based in San Francisco, worries that many of today’s licensing deals are a product of the past funding boom. Without fresh investment, he warns, China’s drug pipeline could run start to run dry.
A bigger concern is rising tensions with America. Because drug prices in the world’s largest economy are less tightly controlled than they are at home, American patients are a big source of revenue for Chinese biotech firms.
American trade restrictions apply to high-tech goods; Chinese biotech has so far escaped similar scrutiny. An attempt to block Chinese firms from supplying some biotech services and equipment has stalled in Congress. But, with Chinese trade firmly in Donald Trump’s sights, biotech may not be able to escape unscathed for much longer.
The approach of selling to America via licensing deals may offer limited protection; already Chinese biotech firms are getting lower licensing fees for their experimental drugs than American counterparts, because of perceived geopolitical risks. Impressive as the pace of Chinese innovation is, it will have to contend with some mighty geopolitical forces. ■

Taiwan US-China expert video: China has surpassed F22, F35 data is fake. The same problems discovered by Elon Musk on USAID lying and faking data, US Military Industrial Complex and the the Pentagon is worst. 台美中美專家影片:中國已超越F22,F35資料造假。馬斯克發現的美國國際開發署說謊和偽造數據,美國軍事工業複合體與五角大廈的關係的腐敗問題更為嚴重
https://rumble.com/v6lhg74-china-has-surpassed-f22-f35-data-is-fake.html
https://www.tiktok.com/t/ZP8YxfSeR/
Full video: https://youtu.be/KqUzEa-aZns
